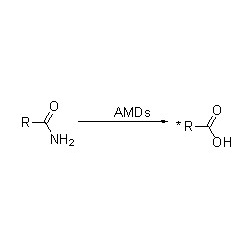
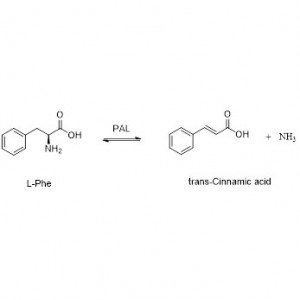
Amidase (AMD)
Enzymes:Su ne macromolecular biocatalists, yawancin enzymes sunadarai ne
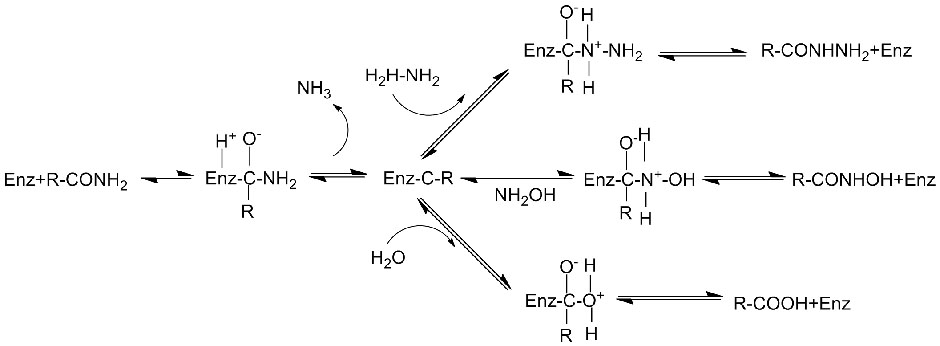
Amidase:Haɓaka hydrolysis na nau'ikan aliphatic da aromatic amides daban-daban na ciki da na waje ta hanyar canja wurin rukunin acyl zuwa ruwa tare da samar da free acid da ammonia. Ana amfani da Hydroxamic acid da sauran Organic acid sosai a matsayin magunguna saboda suna cikin abubuwan da ke haifar da ci gaba, maganin rigakafi da hana ciwace-ciwacen daji. Ana iya raba amidases zuwa nau'in R da S nau'in acylases bisa ga tsarin stereoselectivity na catalyst.
Baya ga ƙarfafa hydrolysis na amides, amidase kuma yana iya haɓaka halayen canja wurin acyl a gaban co-substrates kamar hydroxylamine.
Amidase mai tushe daban-daban yana da takamaiman yanayin substrate daban-daban, wasu daga cikinsu zasu iya hydrolyze aromatic amides kawai, wasu daga cikinsu zasu iya hydrolyze aliphatic amides kawai, wasu kuma zasu iya hydrolyze α-ko ω-amino amides. Yawancin amines suna da kyakkyawan aikin catalytic kawai don acyclic ko sauƙi aromatic amides, amma ga hadaddun aromatics, heterocyclic amides, musamman amides tare da ortho substituents, gabaɗaya suna da ƙarancin aiki (ƙananan enzymes ne kawai ke nuna mafi kyawun tasirin catalytic).
Tsarin haɓaka kuzari:
| Enzymes | Lambar Samfura | Lambar Samfura |
| Foda na Enzyme | ES-AMD-101~ ES-AMD-119 | saitin amidases 19, 50 MG kowanne 19 samfura * 50mg / samfura, ko wani adadi |
| Kayan tantancewa (SynKit) | ES-AMD-1900 | saitin amidases 19, 1 mg kowanne 19 abubuwa * 1mg / abu |
★ Babban takamaiman substrate.
★ Ƙarfin zaɓin chiral.
★ Ingantaccen aiki na juyawa.
★ Rage yawan kayayyakin da aka samar.
★ Yanayin amsawa mai sauƙi.
★ Mai kyau ga muhalli.
➢ Ya kamata a yi gwajin enzyme don takamaiman substrates saboda takamaiman substrates, kuma a sami enzyme wanda ke haɓaka substrates ɗin da aka nufa tare da mafi kyawun tasirin catalytic.
➢ Kada ka taɓa haɗuwa da yanayi mai tsanani kamar: zafi mai yawa, pH mai yawa/ƙasa da kuma sinadarin sinadarai masu ƙarfi waɗanda ke da yawan amfani.
➢ A al'ada, tsarin amsawar yakamata ya haɗa da substrate, maganin buffer (mafi kyawun pH na amsawar enzyme). Ya kamata a sami haɗin gwiwa kamar hydroxylamine a cikin tsarin amsawar canja wurin acyl.
➢ Ya kamata a ƙara AMD a ƙarshe a cikin tsarin amsawa tare da mafi kyawun pH da zafin jiki.
➢ Duk nau'ikan AMD suna da yanayi daban-daban na amsawa mafi kyau, don haka ya kamata a ƙara yin nazari a kansu.
Misali na 1(1):
Ayyukan hydrolysis zuwa nau'ikan amide substrates daban-daban
| Substrate | Takamaiman aiki μmols min-1mg-1 | Substrate | Takamaiman aiki μmols min-1mg-1 |
| Acetamide | 3.8 | ο- OH-benzamide | 1.4 |
| Propionamide | 3.9 | p- OH-benzamide | 1.2 |
| Lactamide | 12.8 | ο-NH2benzamide | 1.0 |
| Butyramide | 11.9 | p-NH2benzamide | 0.8 |
| Isobityramide | 26.2 | ο-Toluamide | 0.3 |
| Pentanamide | 22.0 | p-Toluamide | 8.1 |
| Hexanamide | 6.4 | Nikotinamide | 1.7 |
| Cyclohexanamide | 19.5 | Isonicotinamide | 1.8 |
| Acrylamide | 10.2 | Picolinamide | 2.1 |
| Metacrylamide | 3.5 | 3-Phenylpropionamide | 7.6 |
| Prolinamide | 3.4 | Indol-3-acetamide | 1.9 |
| Benzamide | 6.8 |
An gudanar da aikin a cikin maganin sodium phosphate buffer mai ƙarfin 50mM, pH 7.5, a digiri 70 na Celsius.
| Amides | Hydroxylamine | Hydrazine |
| Acetamide | 8.4 | 1.4 |
| Propionamide | 18.4 | 3.0 |
| Isobityramide | 25.0 | 22.7 |
| Benzamide | 9.2 | 6.1 |
An gudanar da aikin a cikin maganin sodium phosphate buffer mai ƙarfin 50mM, pH 7.5, a digiri 70 na Celsius.
Yawan sinadaran da ke da alaƙa: amides, 100 mM(benzamide, 10 mM); hydroxylamine da hydrazine, 400 mM; enzyme 0.9 μM.
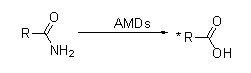
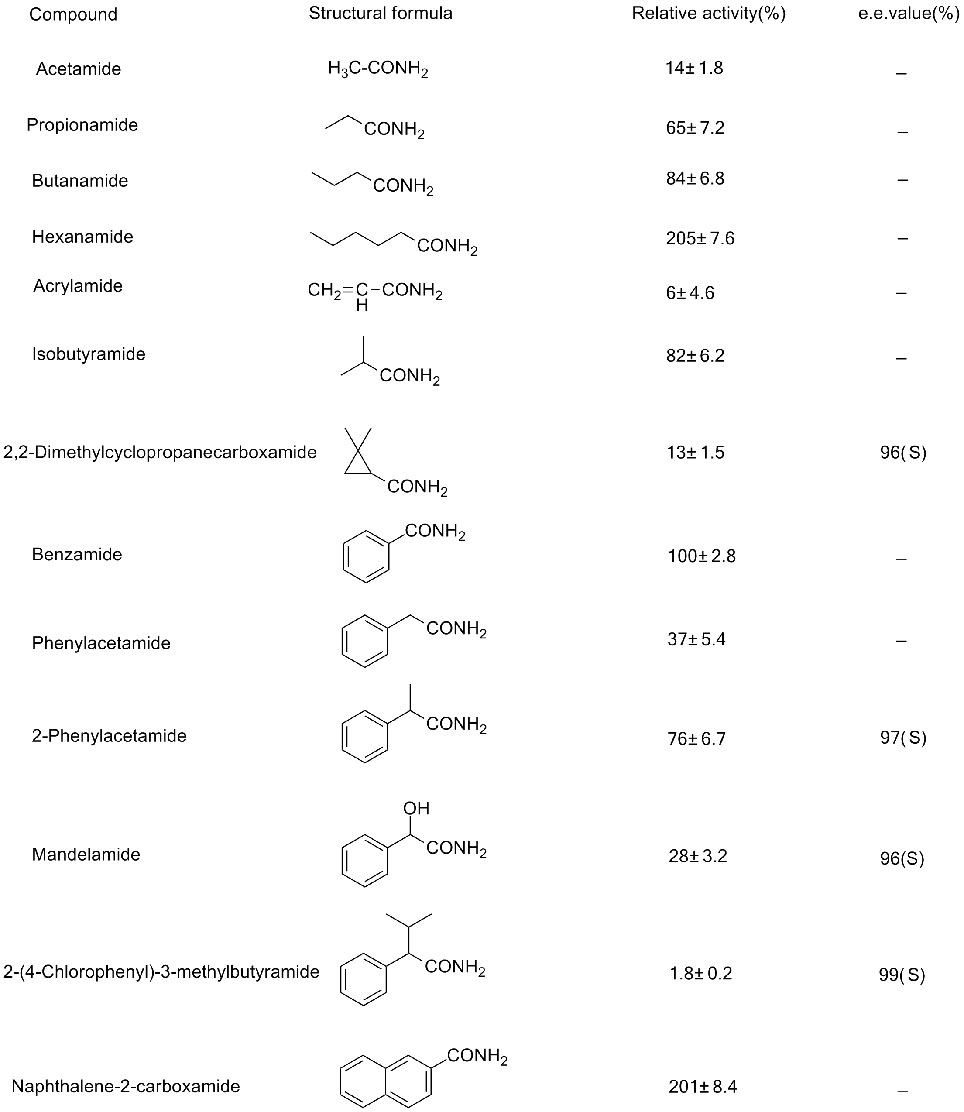
Misali na 2(2):
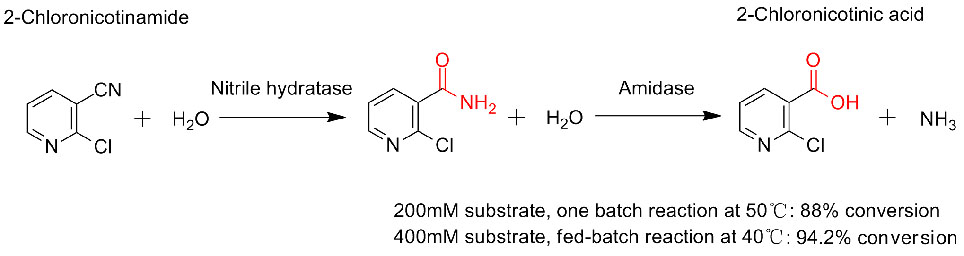
Misali na 3(3):
1. D'Abusco AS, Ammendola S., et al. Extremophiles, 2001, 5:183-192.
2. Guo FM, Wu JP, Yang LR, da sauransu. Process Biochemistry, 2015, 50(8): 1400-1404.
3. Zheng RC, Jin JQ, Wu ZM, da sauransu. Bioorganic Chemistry, 2017, Akwai a yanar gizo 7.